In a recent paper by Solimini et al the authors discuss the
concepts of STOP and GO genes and carcinogenesis[1]. The paper reports on some
extensive experimental results focusing on the issue of proliferation and the
loss of certain sets of gene sites, the STP and GO sites.
The authors begin by discussing the current concepts of
changes in oncogenes and tumor suppressor genes, some of the key pathway
elements that we examine in analyzing intracellular pathway dynamics. They
state:
Cancer progression is directed by alterations in
oncogenes and tumor suppressor genes (TSGs) that provide a competitive advantage
to increase proliferation, survival, and metastasis. The cancer genome is
riddled with amplifications, deletions, rearrangements, point mutations, loss
of heterozygosity (LOH), and epigenetic changes that collectively result in
tumorigenesis.
How these changes contribute to the disease is a central
question in cancer biology. In his “two-hit hypothesis,”
Knudson proposed that two mutations in the same gene are required for
tumorigenesis, indicating a recessive disease. In addition, there are now
several examples of haploinsufficient TSGs .
Current models do not explain the recent observation that
hemizygous recurrent deletions are found in most tumors. Whether multiple genes
within such regions contribute to the tumorigenic phenotype remains to be elucidated…
The last sentence regarding the inability to explain the
presence of hemizygous deletions under the current model is the main driver for
this effort. Thus they argue and demonstrate experimentally that:
Tumors exhibit numerous recurrent hemizygous focal
deletions that contain no known tumor suppressors and are poorly understood. To
investigate whether these regions contribute to tumorigenesis, we searched
genetically for genes with cancer-relevant properties within these hemizygous
deletions.
We identified STOP and GO genes, which negatively and
positively regulate proliferation, respectively.
STOP genes include many known tumor suppressors, whereas
GO genes are enriched for essential genes.
Analysis of their chromosomal distribution revealed that
recurring deletions preferentially over-represent STOP genes and under-represent
GO genes.
We propose a hypothesis called the cancer gene
island model, whereby gene islands encompassing high densities of STOP
genes and low densities of GO genes are hemizygously deleted to maximize
proliferative fitness through cumulative haploinsufficiencies.
Because hundreds to thousands of genes are hemizygously deleted
per tumor, this mechanism may help to drive tumorigenesis across many cancer
types.
This is an intriguing hypothesis. It adds more pieces to an
already complex puzzle. The Cancer Gene Island, CGI, hypothesis seems to
indicate the complex changes in multiple gene sites. In particular there was a
deletion of the STOP genes in preference to the GO genes. Unfortunately there
did not seem to be a mechanism for these deletions, however the experimental
evidence does indicate the phenomenon.
In their experimental analysis they have observed certain in
vitro results which compel their hypothesis. They state:
This in silico analysis suggests that the loss of a
single copy of GO genes has a negative impact on cellular fitness. To
independently test this hypothesis, we turned to the other arm of our screen
that identified candidate GO genes whose depletion limits proliferation and
survival. Because both normal and cancer cells are dependent on these essential
GO genes, we analyzed data from proliferation screens on HMECs, one normal
prostate epithelial cell line, and seven breast or prostate cancer cell lines
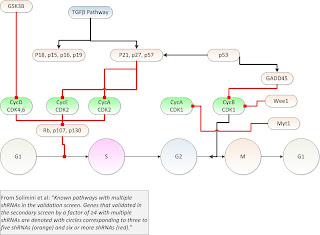
They provide an interesting pathway model as shown below (as
mofified, and also not that they have short hairpin RNAs (shRNAs)).
They conclude as follows:
The enrichment for genes localized to deletions suggests
that we have identified dozens of new TSGs in recurrent deletions. We have also
likely identified more TSGs outside of these regions because the STOP gene set
is (i) enriched for known TSGs, many of which are not found in recurrent
deletions, and (ii) enriched for genes that undergo somatic loss-of-function
mutation.
Finally, this work suggests that cells possess a substantial
number of genes that restrain proliferation in vitro, which could be
inactivated to promote clonal expansion during tumorigenesis in addition to the
traditional driver genes currently known. Given the prevalence of multiple,
large, recurring hemizygous deletions encompassing skewed distributions of
growth control genes in tumors, we propose that the elimination of cancer gene
islands that optimize fitness through cumulative haplo-insufficiencies may play
an important role in driving tumorigenesis, with implications for the way in
which we think about cancer evolution.
As with many such works this raises as many questions as it
seems to answer. However the control or lack thereof of proliferation and the
cell cycle is a critical issue in carcinogenesis.
[1] Solimini, N., et al, Recurrent Hemizygous Deletions in
Cancers May Optimize Proliferative Potential, Science, 6 JULY 2012 VOL 337, p
104.